System Maintenance occurs every Friday.
A TEM (transmission electron microscope) uses a highly energetic electron beam (100 keV - 1 MeV) to image and obtain structural information from thin film samples. The electron microscope consists of an electron gun, or source, and an assembly of magnetic lenses for focusing the electron beam. Apertures are used to select among imaging modes and to select features of interest for electron diffraction work. The sample is illuminated with an almost parallel electron beam, which is scattered by the sample. In crystalline materials, the scattering takes the form of one or more Bragg diffracted beams, which are used to form a transmission diffraction pattern. These diffraction patterns can be used to identify unknown phases in the sample. A bright-field image of the sample can be formed by looking at the straight-through, non-diffracted beam. Features in the sample that cause scattering have darker contrast in a bright-field image than those that cause little or no scattering. An electron diffraction pattern can be generated from a particular area in a bright-field image (such as a particle or grain) by using a selected area aperture. Dark-field images are formed from a single diffracted beam and are used to identify all the areas of a particular phase having the same crystalline orientation. Magnifications from about 100x up to several hundred thousand times can be achieved in the TEM.
After the theory of the wave nature of electrons was developed by Louis DeBroglie in 1925, researchers Knoll and Ruska proposed the idea of an electron microscope. In 1936, the first electron microscope was built by Metropolitan-Vickers in the U.K. After World War II, several manufacturers, including Hitachi, JEOL, Philips, and RCA began manufacturing electron microscopes. In the late 1940's, Heidenreich first thinned specimens to electron transparency. Hirsh and his colleagues at Cambridge then developed the theory of electron diffraction contrast and documented his work in a large volume sometimes referred to as the "TEM Bible." The use of TEM for materials science work and semiconductor analysis has increased rapidly in the past two decades. This trend is likely to continue as feature sizes shrink. The TEM is probably one of the best positioned tools for examination of semiconductor structures for the foreseeable future.
Electron Optics
There are basically three types of electron guns on today's transmission electron microscopes: 1. the tungsten cathode, 2. the lanthanum hexaboride (LaB6) cathode, and 3. the field emission gun. The three electron guns are described briefly below.
1. Tungsten cathode
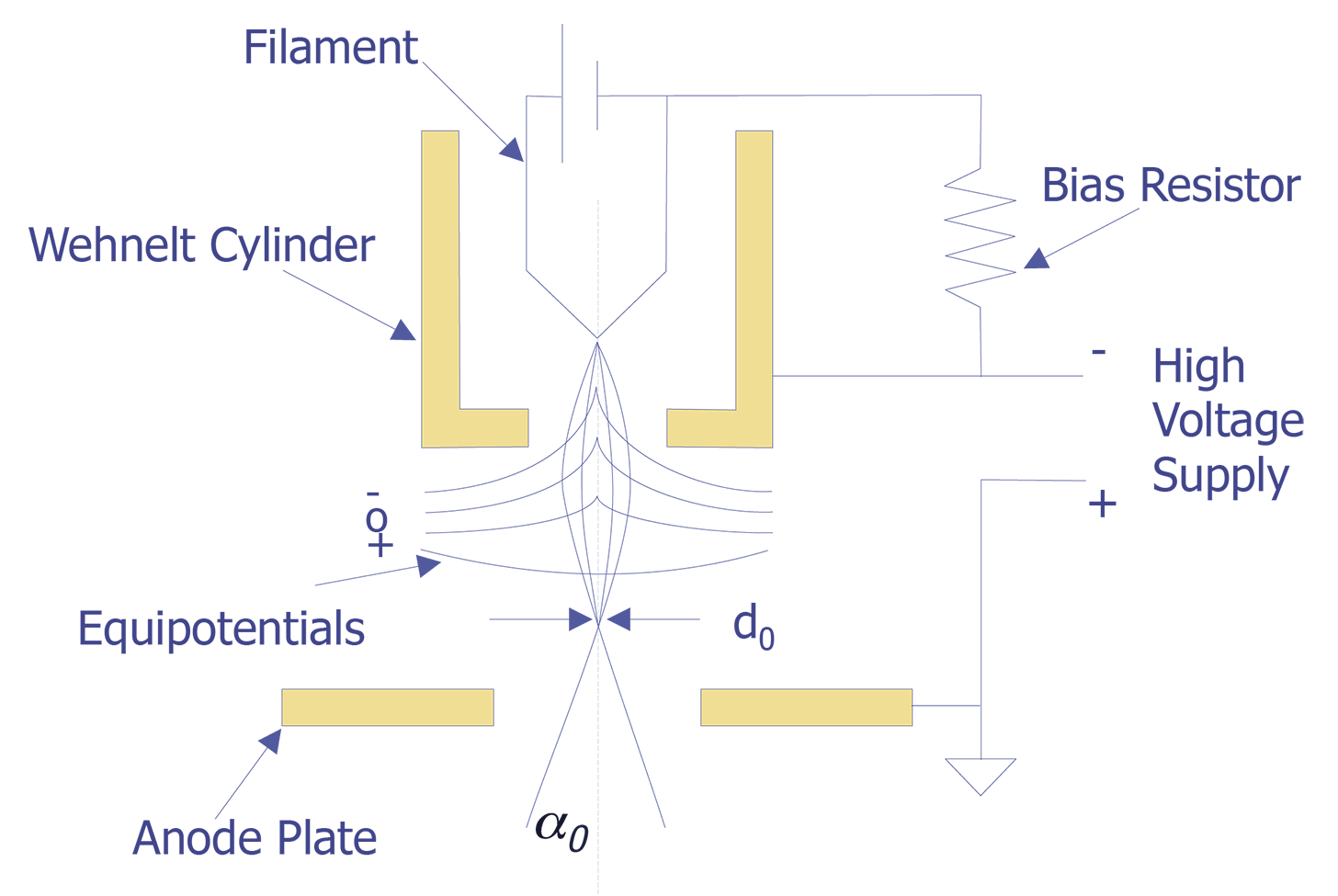
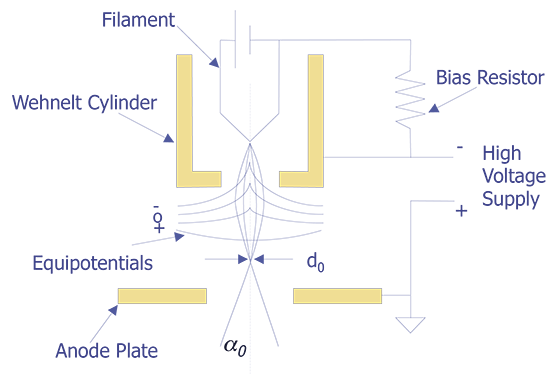
The tungsten cathode is a fine wire approximately 100mm in diameter that has been bent into the shape of a hairpin with a V-shaped tip. The tip is heated to around 2400°C by passing current through it. At this temperature, one can expect a current density of approximately 1.75 A/cm2. The electrons will have a potential distribution of 0 to 2 volts. With a bias voltage between 0 and 500 volts, the electrons can be accelerated toward the anode. At an emission of 1.75 A/cm2, the lifetime of the tungsten filament is approximately 50 hours. A schematic diagram of a tungsten cathode is shown below (see Figure 1).
2. Lanthanum hexaboride (LaB6) cathode
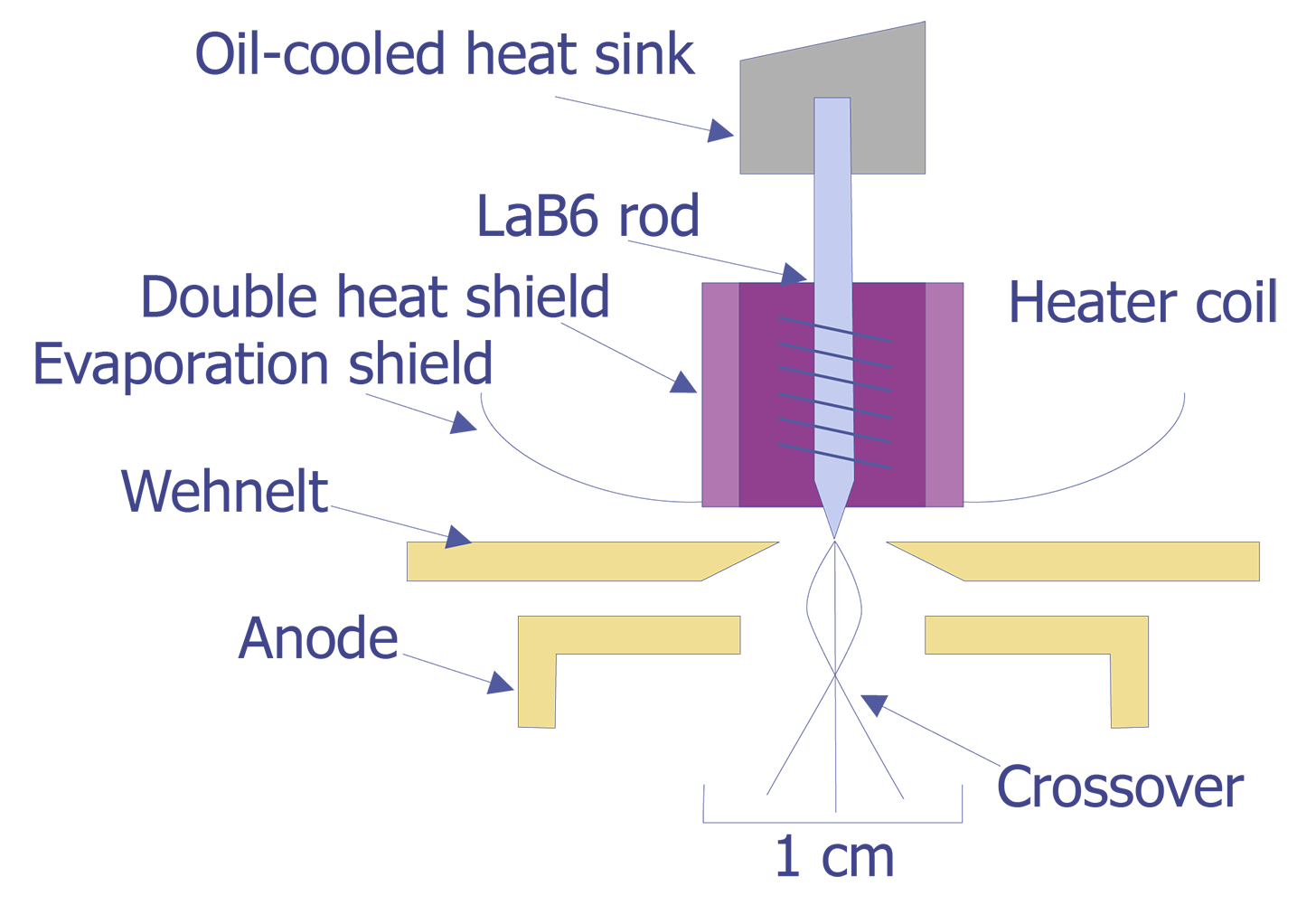
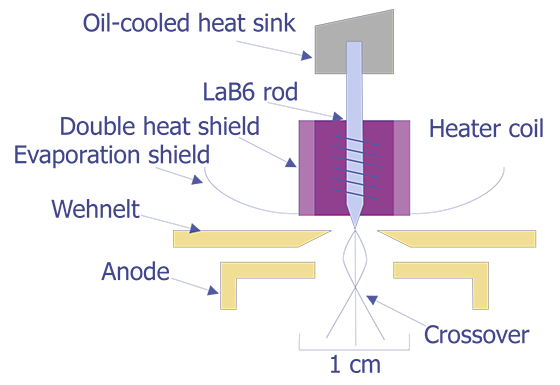
As the need for higher resolution imaging increased, so did the need for brighter filaments. The most straightforward method to achieve this goal is to find a material with a lower work function Ew. A lower work function means more electrons at a given temperature, hence a brighter filament and higher resolution. Lathanum hexaboride, commonly known as LaB6, has been the best material developed to date for this application. The LaB6 filament operates at approximately 2125°C, resulting in a brightness on the order of five times brighter than a tungsten filament under the same conditions. LaB6 filaments tend to be an order of magnitude more expensive than tungsten filaments. A schematic of the LaB6 filament is shown in Figure 2.
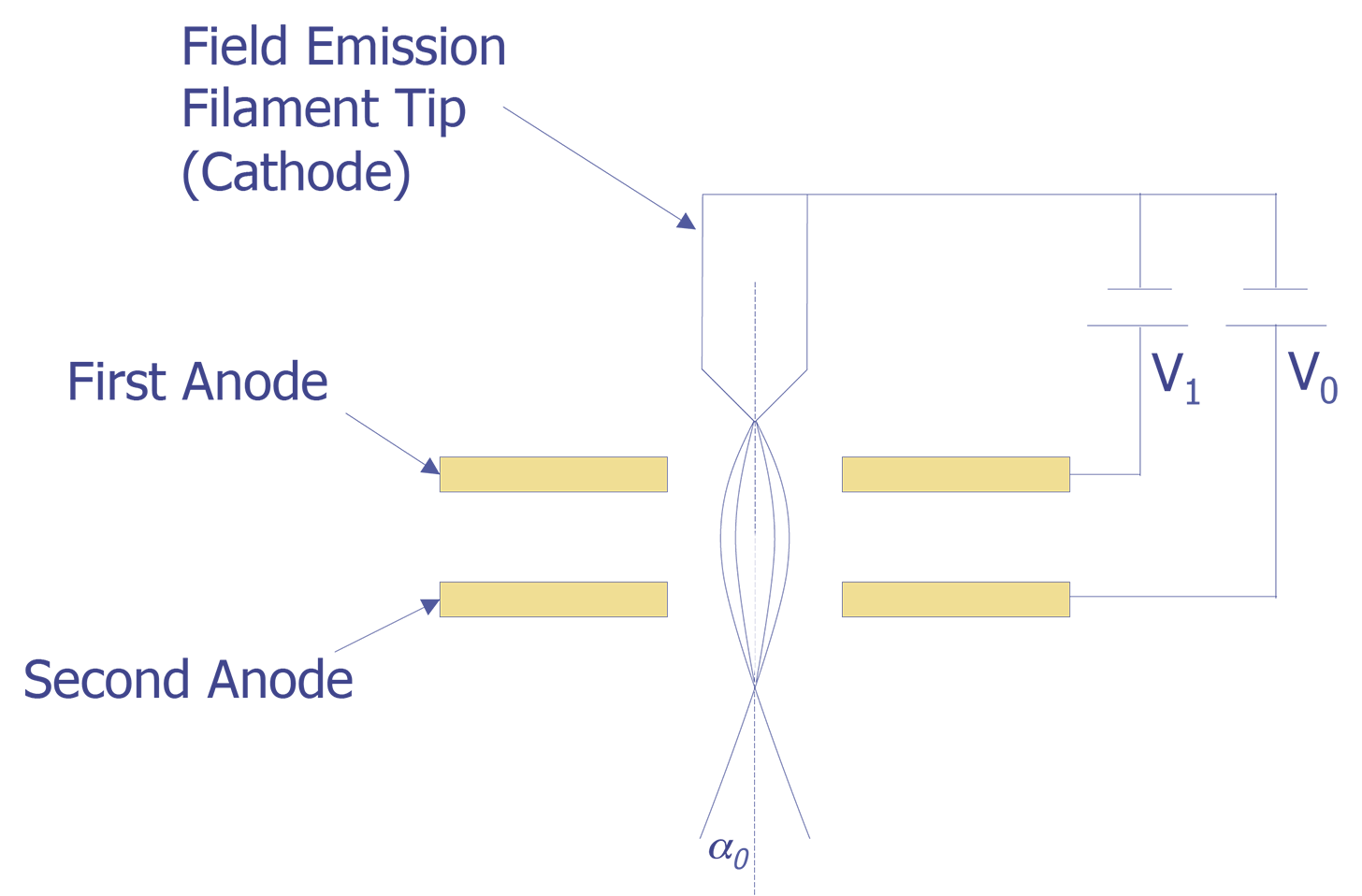
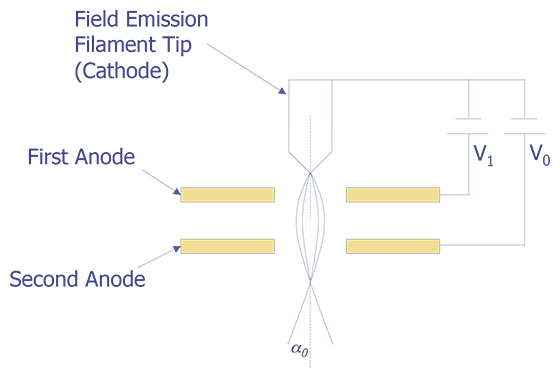
3. Field Emission Gun
Another method for generating electrons is the field emission gun. When the cathode forms a very sharp tip (typically 100 nm or less) and the cathode is placed at a negative potential with respect to the anode, so that the local field at the tip is very strong (greater than 107 V/cm), electrons can tunnel through the potential barrier and become free. Although the total current is lower than either the tungsten or the LaB6 emitters, the current density is between 103 and 106 A/cm, making it hundreds of times brighter than a thermionic emission source. Furthermore, since the electrons are field-generated rather than thermally generated, the tip remains at room temperature. Tips are usually made from tungsten, etched in the <111> plane to generate the lowest work function. Because a native oxide will quickly form on the tip even at moderate vacuum levels (10 mPa), a high vacuum system (10 nPa) is needed. To keep the tip diameter sufficiently small, the cathode can be kept warm (800-1000 °C ) or rapidly heated to approximately 2000F °C for a few seconds to blow off material. A schematic of a field emission tip is shown in Figure 3.
Electron Lenses
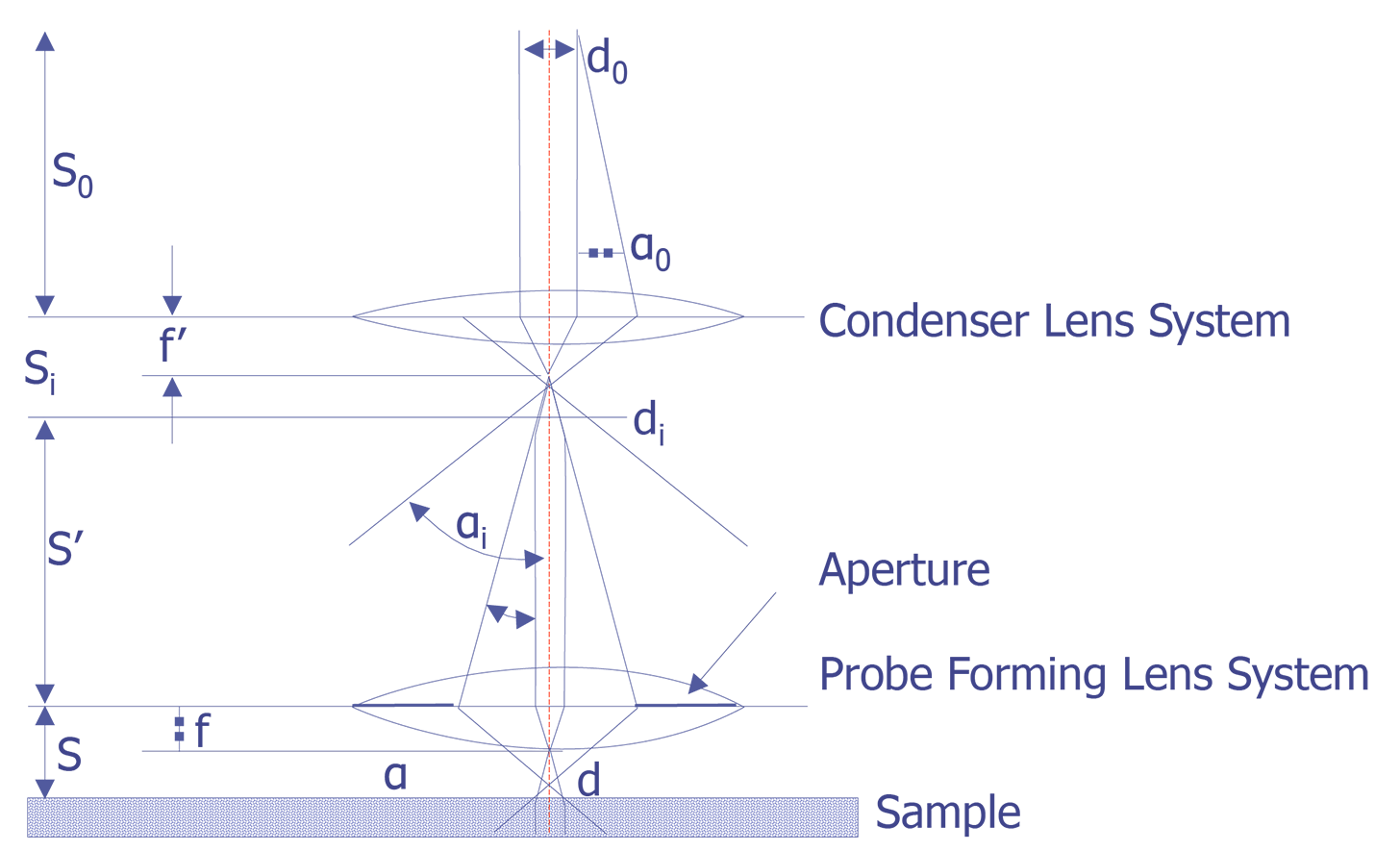
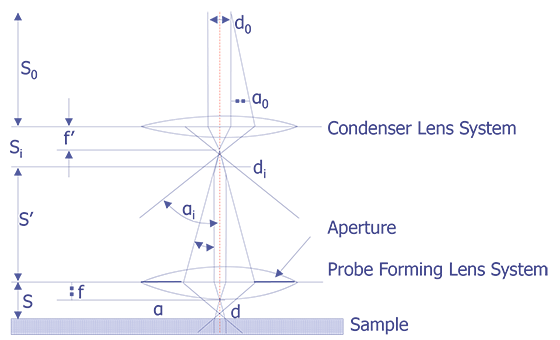
The electron beam is focused using electromagnetic lenses. The condenser lens system, which is composed of one or more lenses, determines the beam current that impinges on the sample. The probe-forming lens, often called the objective lens, determines the final spot size of the electron beam. Conventional electromagnetic lenses are used and the electron beam is focused by the interaction of the electromagnetic field of the lens on the moving electrons. A schematic depiction of the scanning electron microscope column is shown below (see Figure 4). The a angles drawn in Figure 4 are exaggerated on purpose to more clearly show the effect of the lenses on the electron optics. The probe lens can be adjusted to focus the beam depending on the working distance.
The working distance is defined as the distance between the bottom pole piece of the objective lens and the sample surface. It is typically 5 to 25 mm. As the working distance is increased, the spot size increases on the sample, given the same final aperture lens. There is a second tradeoff between spot size and beam current. The spot size is directly controlled by minimizing di (see Figure 4). However, by minimizing di, the beam current falls off by the ratio (aa/ai)2. The operator must make a conscious decision as to whether the analysis requires minimizing the beam spot size or maximizing the beam current.
Several aberrations can occur in an electron optical column. These include: spherical aberration, chromatic aberration, diffraction, and astigmatism. The first three are depicted schematically in Figure 5. Spherical aberration results from non-uniformity of the lenses. Electrons which pass through the lens further off the optical axis are pulled more strongly than those that pass through near the center of the lens. To reduce this effect, the final aperture can be reduced, but this results in a lower beam current.
Chromatic aberration results from differences in electron velocity through the lenses. Electrons with higher velocity or energy will be bent more strongly by the magnetic lenses, resulting in a dull or blurred image. Other than more expensive LaB6 or field emission instruments, no other techniques exist to address this problem. Tungsten filament systems typically have a 2 eV spread when leaving the cathode; LaB6 systems have a 1 eV spread, and field emission systems have a 0.2 to 0.5 eV spread. Diffraction occurs because of the wave nature of electrons and the aperture size of the final lens. The only way to reduce diffraction problems is to increase the final aperture size. Astigmatism results from the fact that magnetic lenses do not have perfect symmetry. Machining errors can cause lens systems to be slightly elliptical, rather than perfectly circular, and irregularities in the iron windings can cause variations in the magnetic field. Most instruments contain a stigmator in the final lens system to help compensate for this effect. The stigmator usually has two controls, one to correct for the magnitude of the asymmetry and one to correct for the direction of the asymmetry of the main field. The stigmator can only correct for asymmetry in the final lens. It cannot correct dirty aperture or filament misalignment.
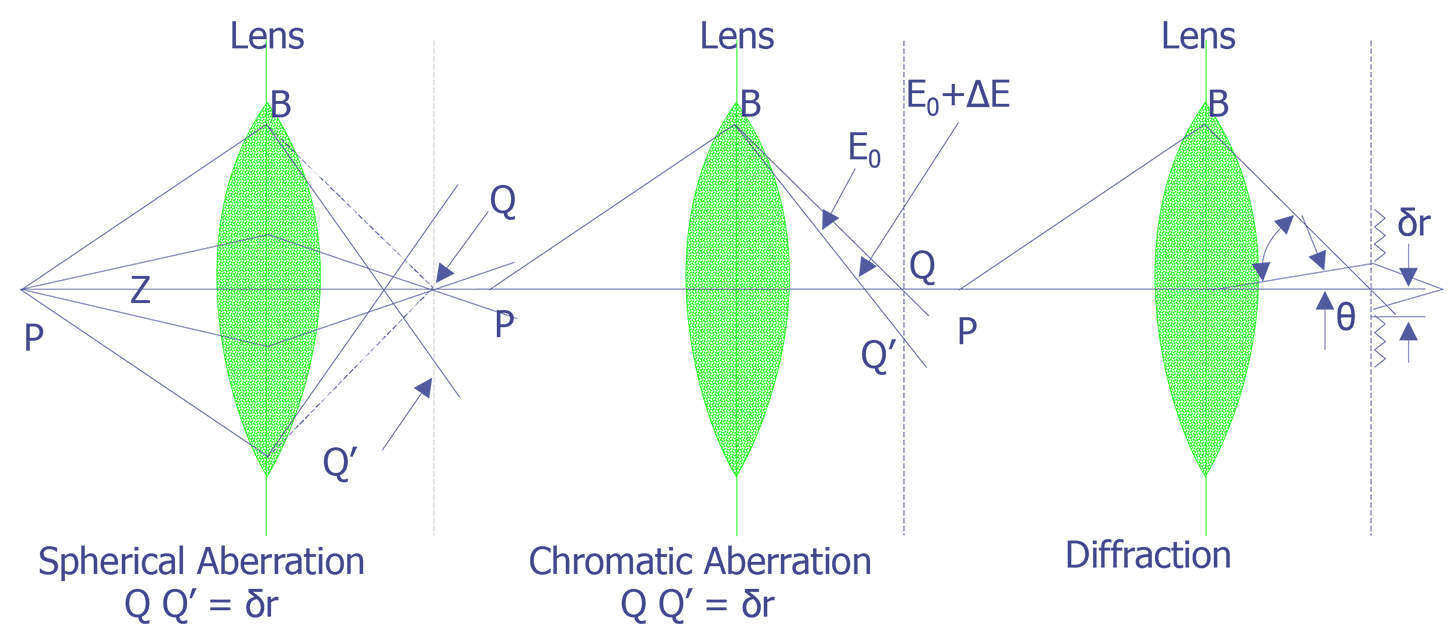
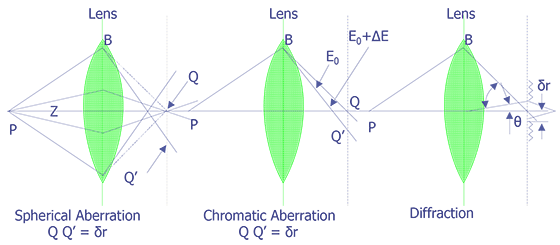
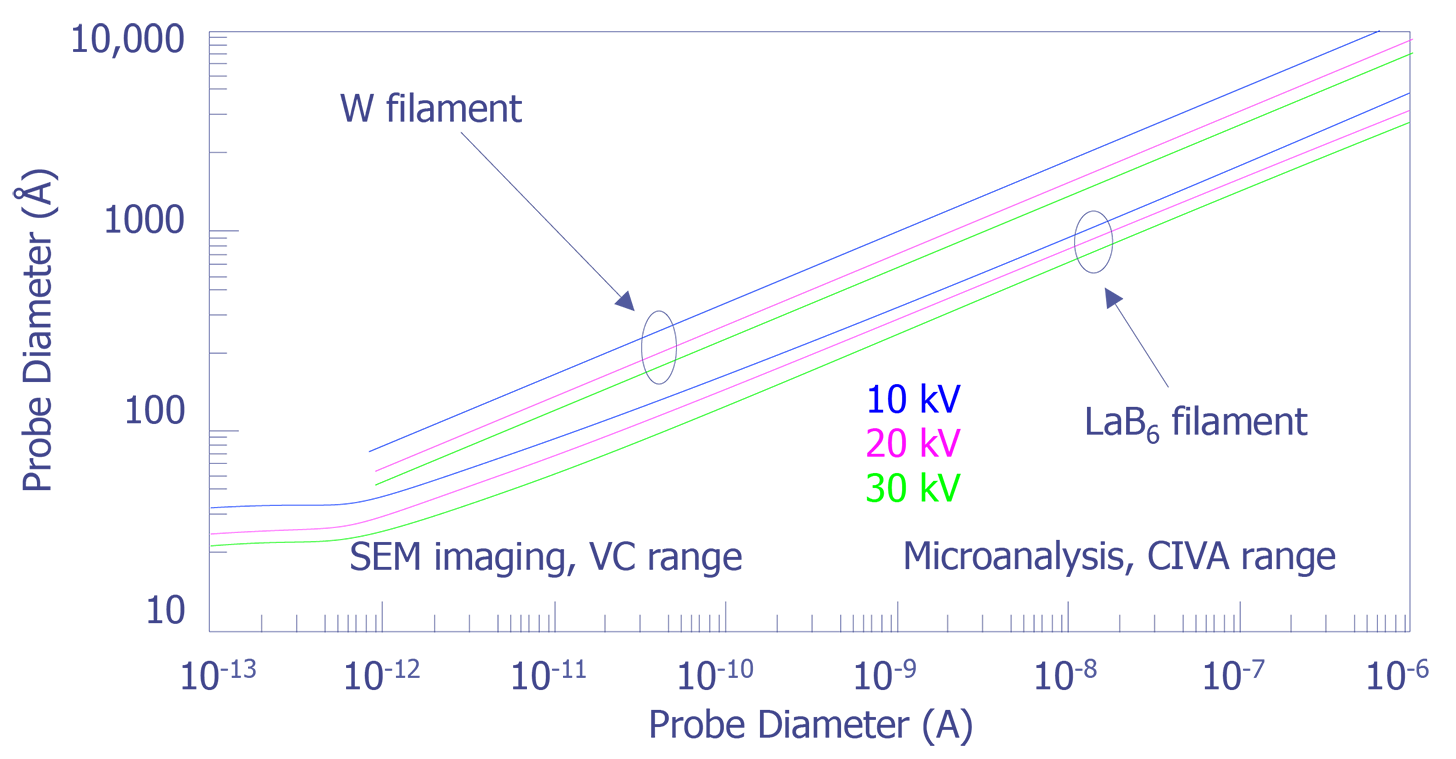
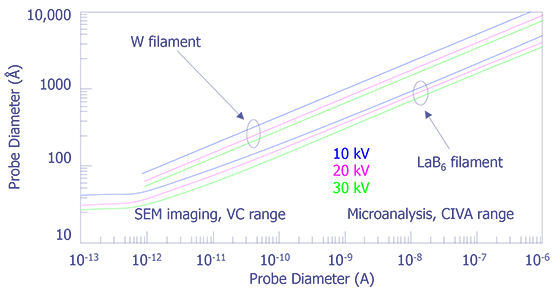
Figure 6 shows the relationship between probe current and beam diameter for both tungsten and LaB6 systems. These values were taken with the tungsten gun current density at 4.1 A/cm2 and the LaB6 gun current density at 25 A/cm2.
There are several basic reasons for using a transmission electron microscope: resolution, depth of field, ability to identify elements, and ability to determine crystalline structure.
According to classical Rayleigh criteria, the smallest distance that can be resolved, δ, is given approximately by
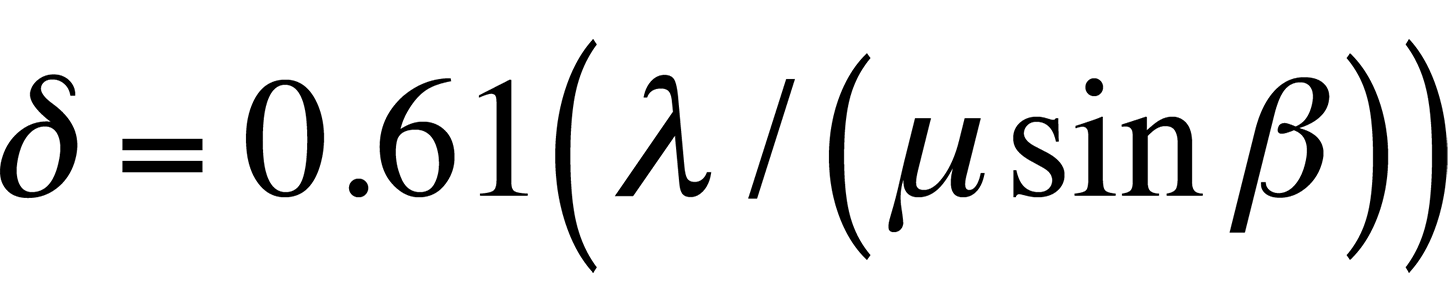
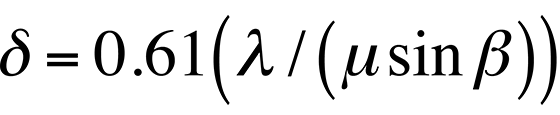
where λ is the wavelength of the radiation, μ is the refractive index of the viewing medium, and β is the semiangle of collection of the magnifying lens. DeBroglie's equation shows that the wavelength λ of electrons is related to their energy, E, by the following equation
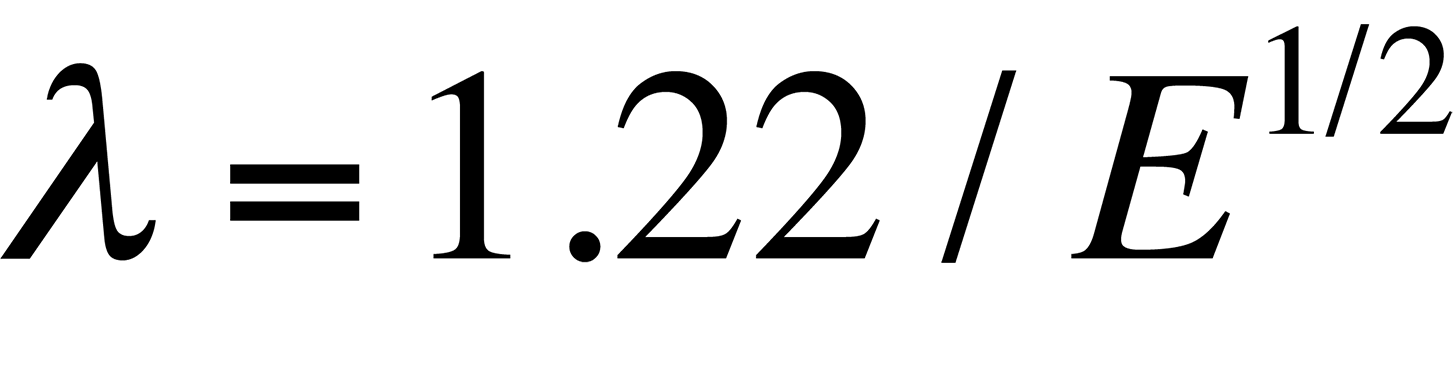
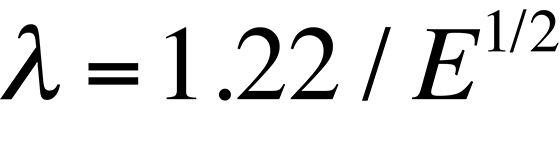
Based on these two equations, a TEM imaging a sample with 100 keV electrons is capable of a theoretical resolution of 0.004 nm (much smaller than the diameter of an atom). In reality, this resolution is not achieved because of our inability to make a "perfect" electron lens. However, the resolution of the TEM is far greater than that of an optical microscope, which is approximately 300 nm for a microscope using visible wavelengths of light.
Because of the intensity of the electron source, we can place a small aperture in front of an electron source, we can dramatically increase the depth of field. A light microscope must have a large aperture in order to capture enough light to make an image. This in turn creates a very small depth of field.
Because the electron beam interacts with elements in the specimen, we can obtain information on the specimen based on signals generated from the interaction. For example, we can use the characteristic x-rays generated to perform energy dispersive x-ray analysis, or use the energy from the inelastically scattered electrons to perform electron energy-loss spectrometry (EELS). We can also use information from elastically scattered electrons to create electron diffraction patterns that can yield data as to the crystalline structure of the specimen.
Even though the transmission electron microscope has several advantages over other instruments, there are a number of factors that make the technique more difficult to use. These include: expertise, expense of the instrument, and sample preparation.
Although the operation of modern transmission electron microscopes is fairly straightforward, the interpretation of the images and spectrometry data requires expertise. Typically, a combination of education in physics and experience in interpreting TEM data is necessary. Many companies may employ a TEM expert. In any case, it is necessary to have a full-time operator.
Transmission Electron Microscopes are relatively expensive instruments. New systems currently cost between $500k and $1M (U.S.). This is more than standard scanning electron microscopes and considerably more than optical microscope systems.
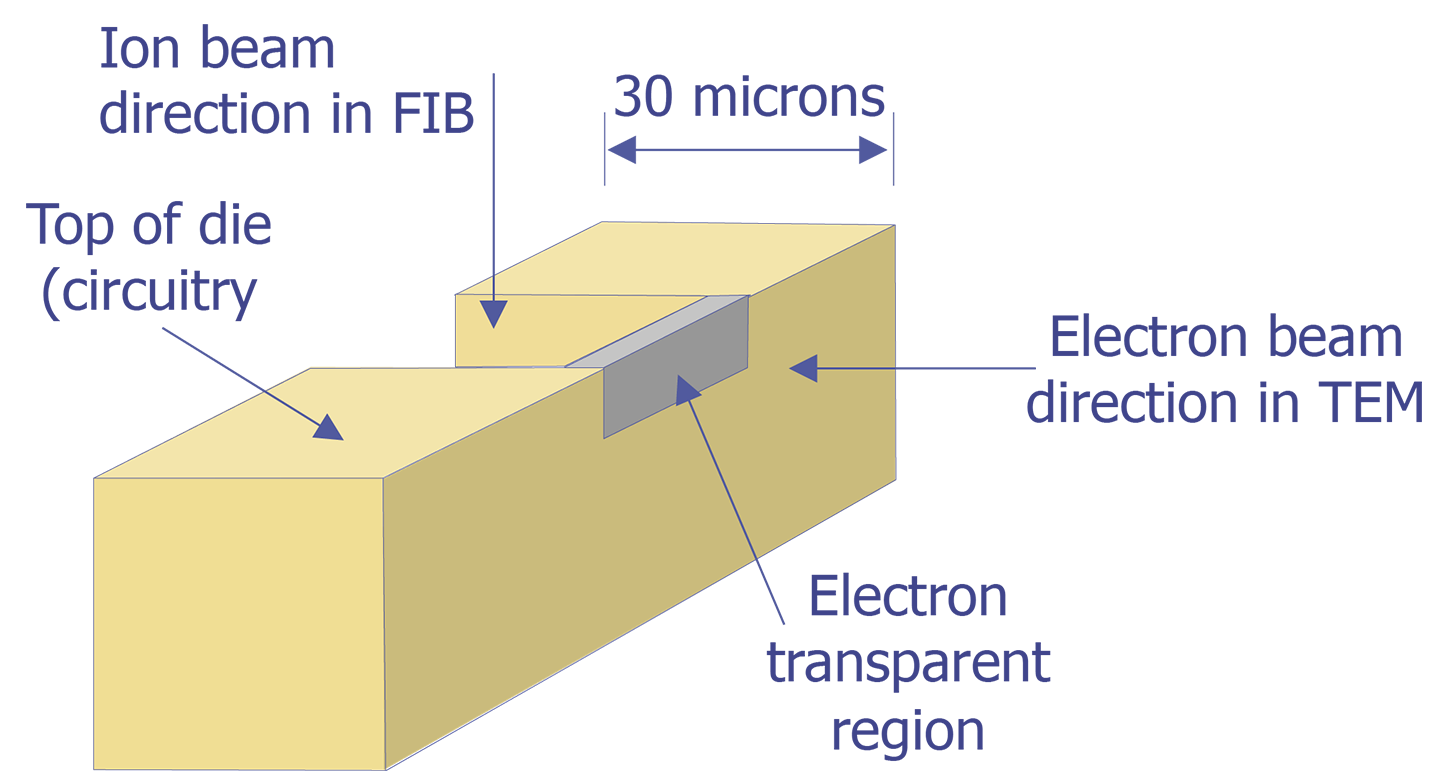
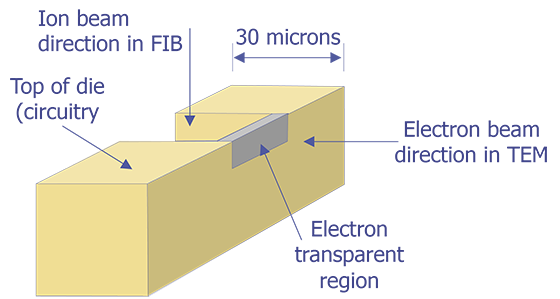
TEM sample preparation is still very much an art. The ability to polish a specimen to electron transparency evenly over several tens of microns takes a good deal of practice and patience. The advent of the focused ion beam is making TEM sample preparation easier, but a focused ion beam system is a very expensive instrument to be routinely used for this purpose.
A TEM is used to obtain structural information about a sample. TEM imaging is used routinely to examine thinned cross-sections of ICs and electronic devices to observe metal and polysilicon grain structure, crystalline defects in silicon (such as stacking faults and dislocations), diffusion barriers, thin metallization layers, defects in gate oxide layers, etc. TEM examination can reveal layer thicknesses and step coverage, show implanted regions, and provide delineation of p/n junctions. Because TEM sample preparation is fairly laborious, TEM analysis is generally reserved for solving those problems that can't be handled with other tools, such as the SEM.
Because TEM sample preparation is destructive, TEM analysis should be performed only after all other analyses have been performed on an IC. Some of the information obtained from TEM analysis can also be obtained from SEM examination of focused ion beam (FIB) or mechanically cross-sectioned devices. These alternative methods should be used whenever possible because of the comparative ease of sample preparation.
Transmission electron microscopes are complex pieces of equipment, and proper training is required in order to operate one. Unless you are a skilled TEM operator yourself, a TEM expert should perform this analysis for you. Your role will be to communicate to the operator what information you need to acquire from the sample, what areas need to be examined, and the like.
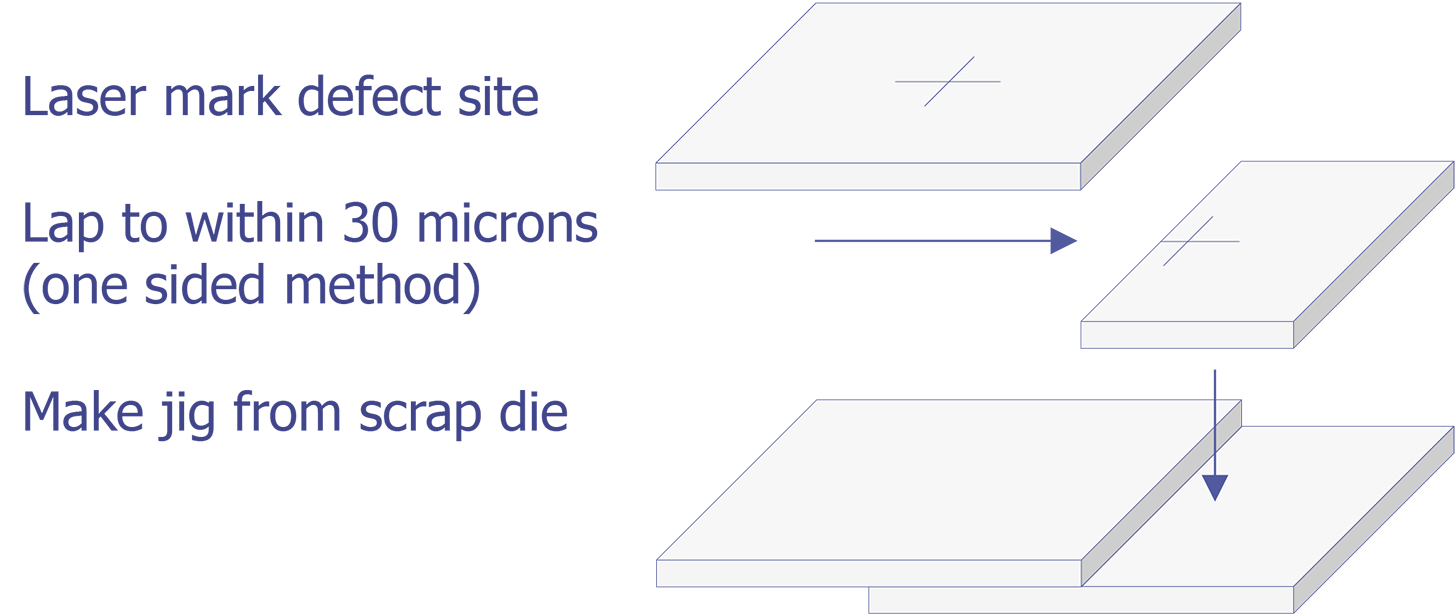
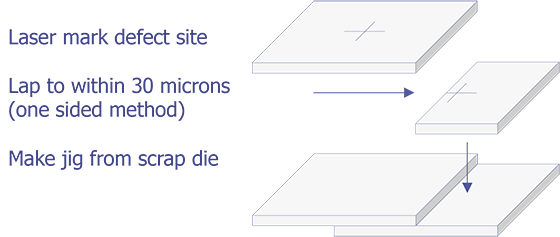
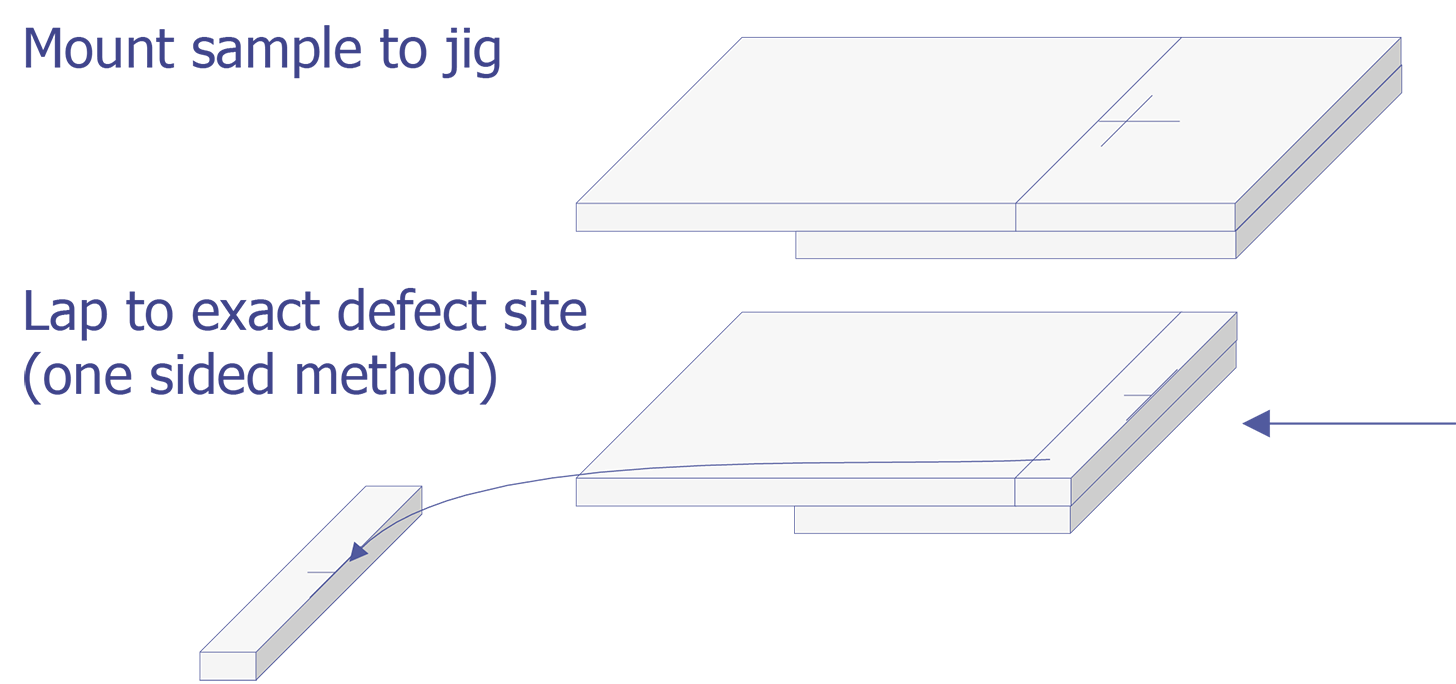
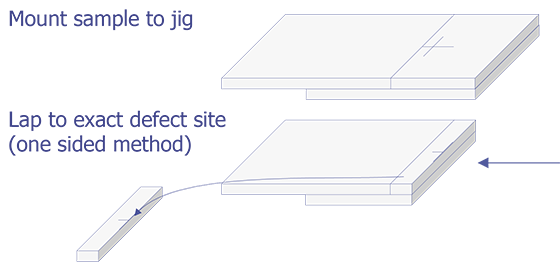
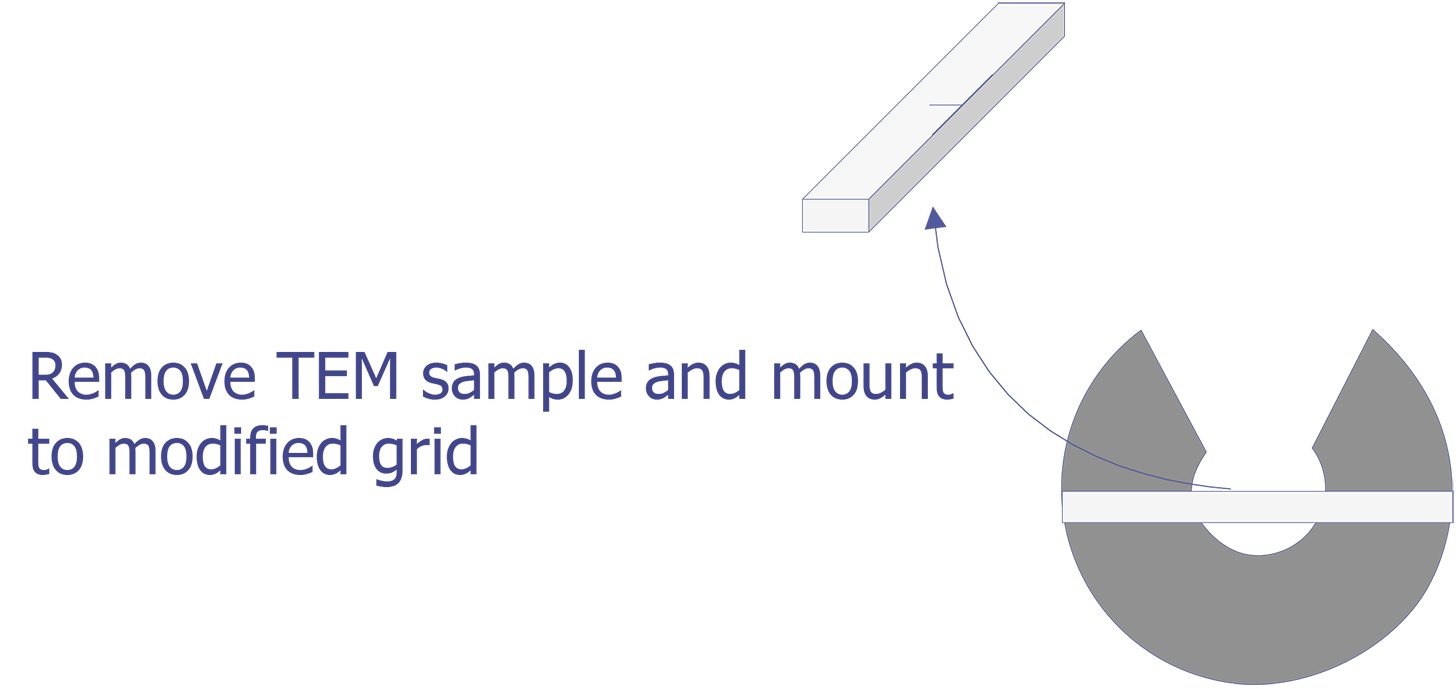
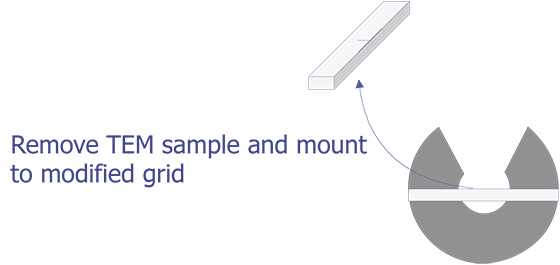
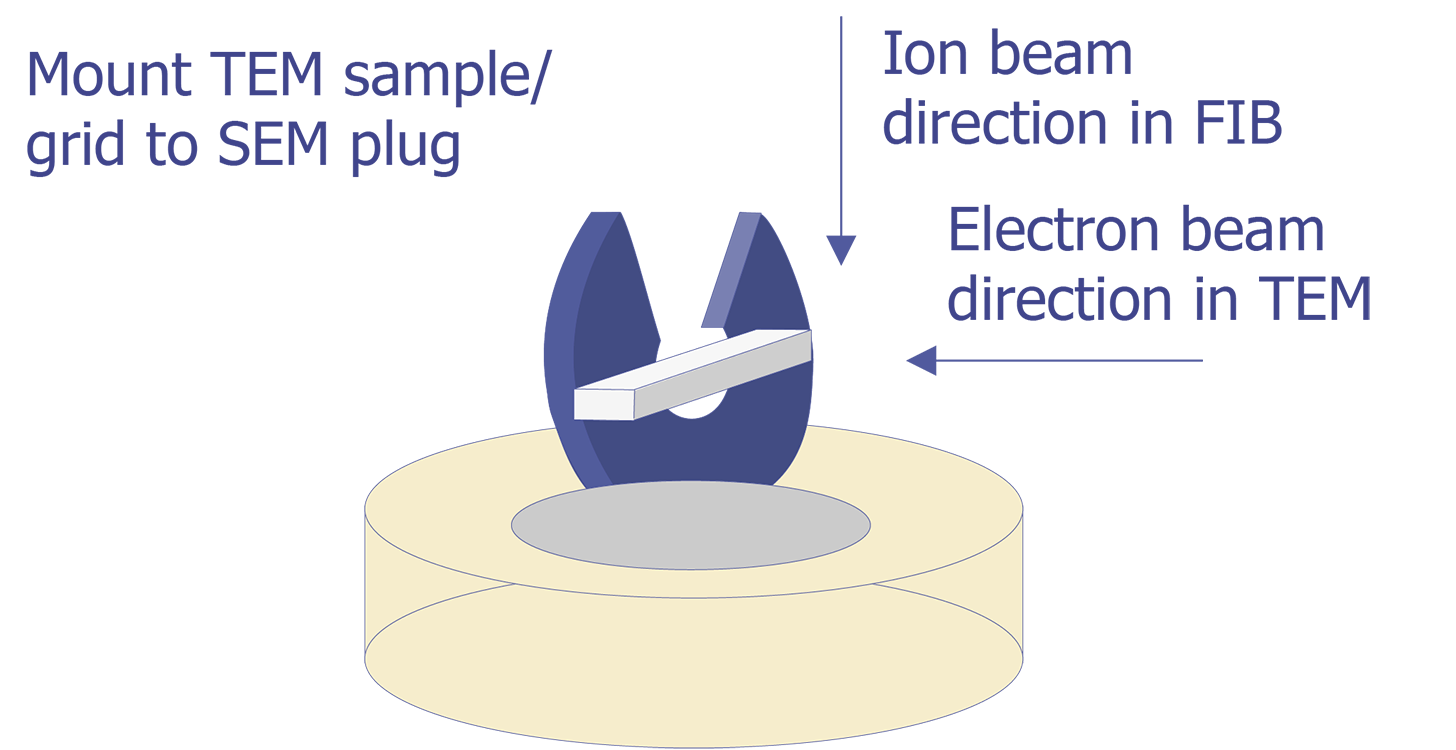
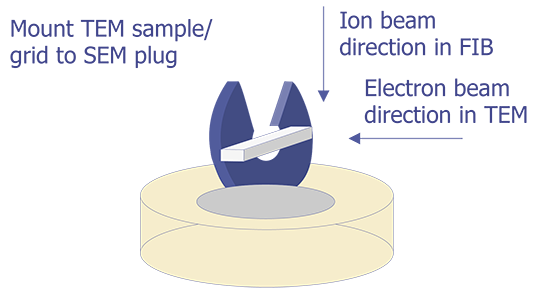
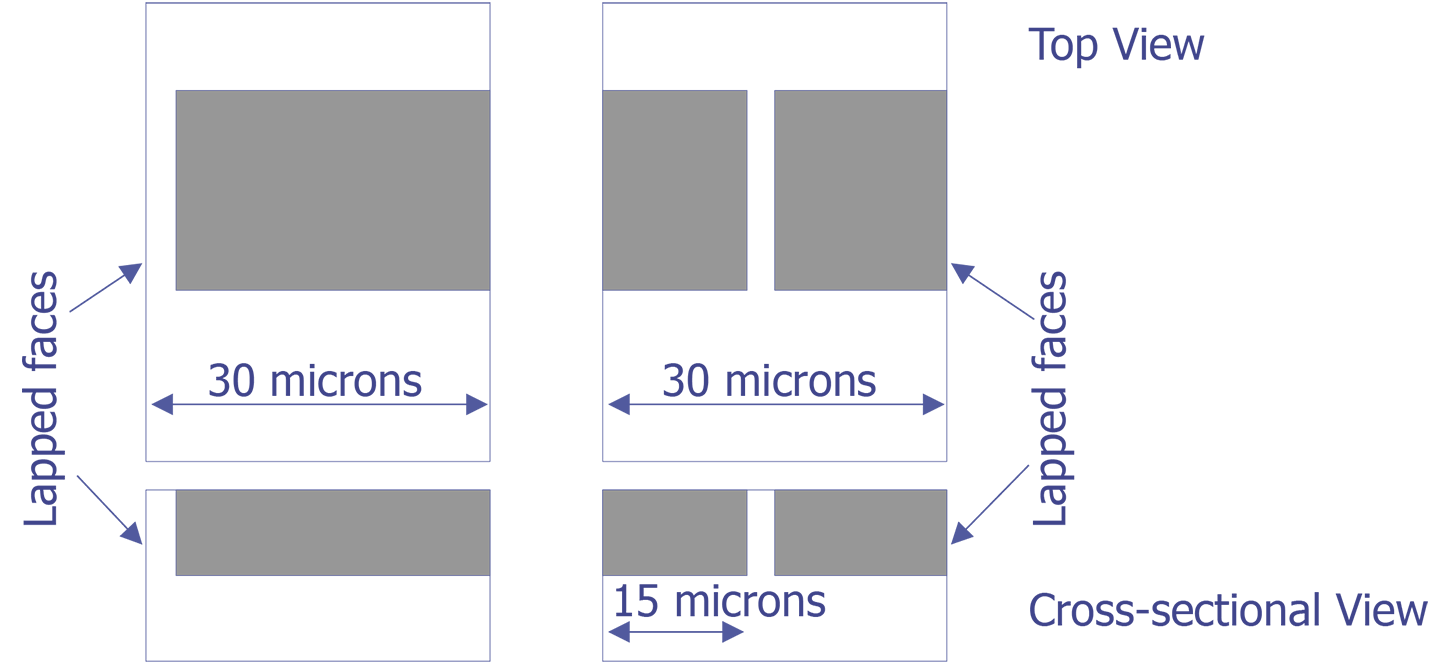
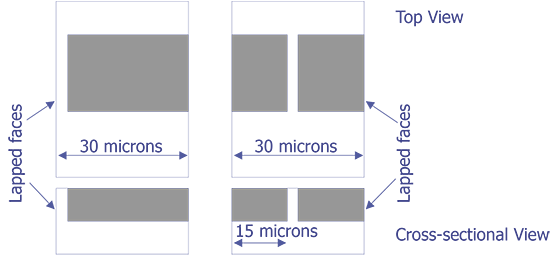
TEM sample preparation for most integrated circuit applications is a two-step procedure that first involves polishing and then ion milling. To prepare a sample for TEM imaging, use the following procedure:
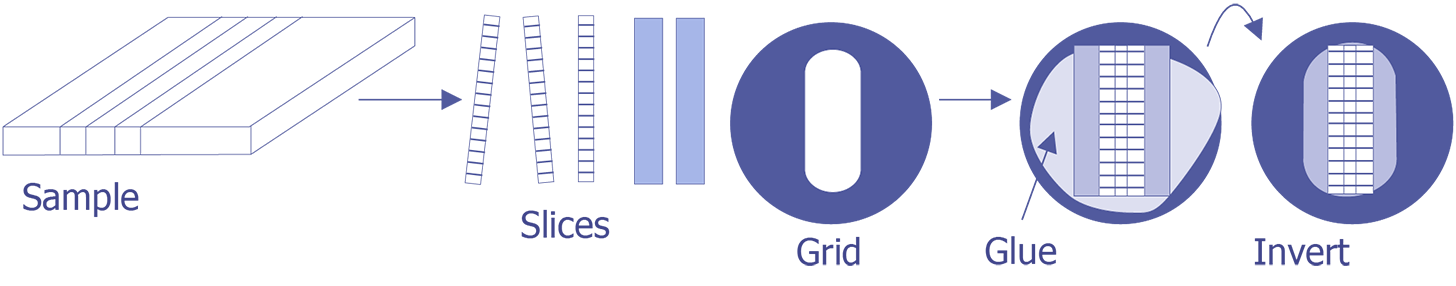
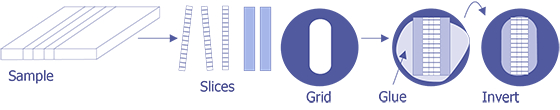
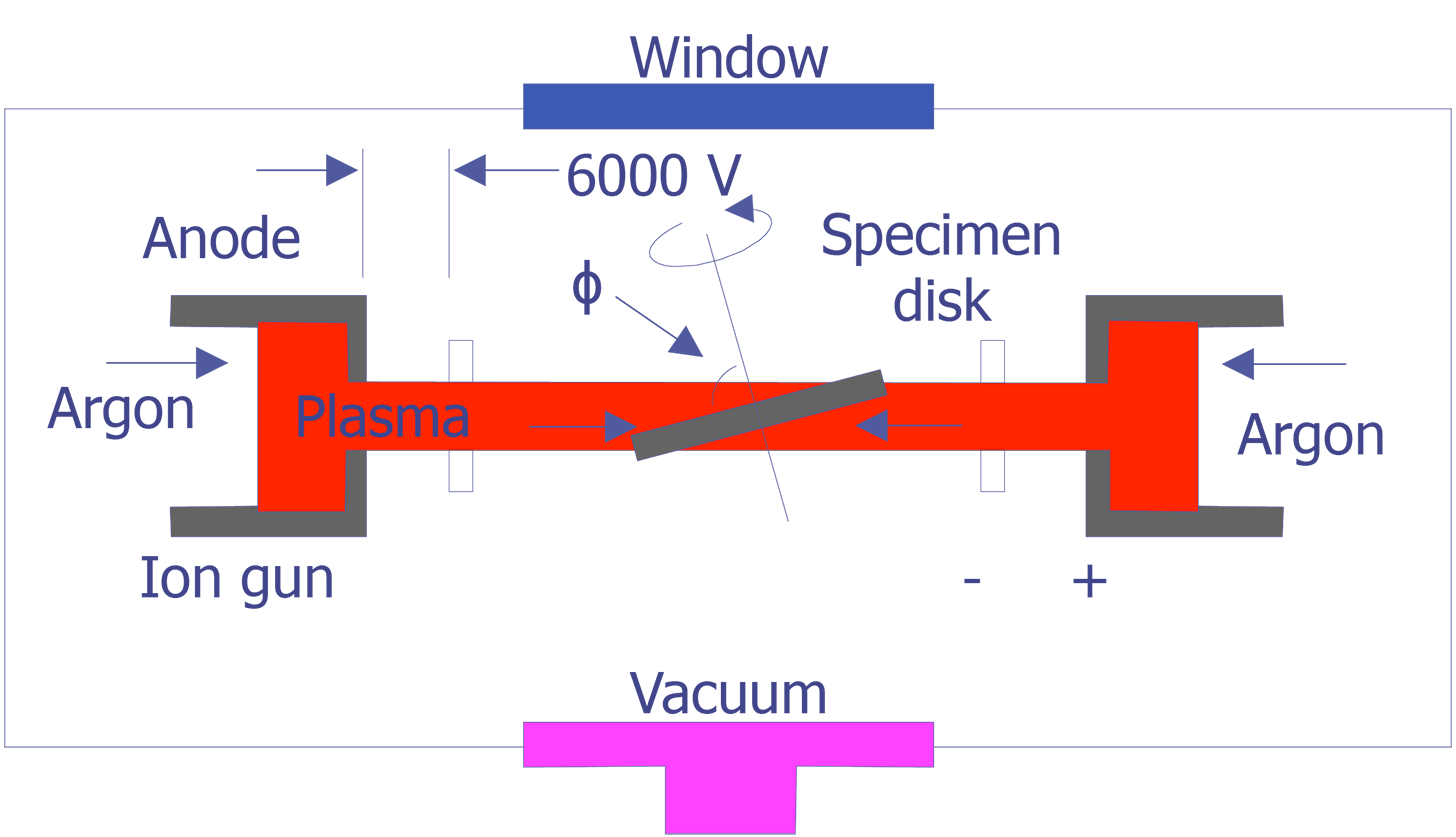
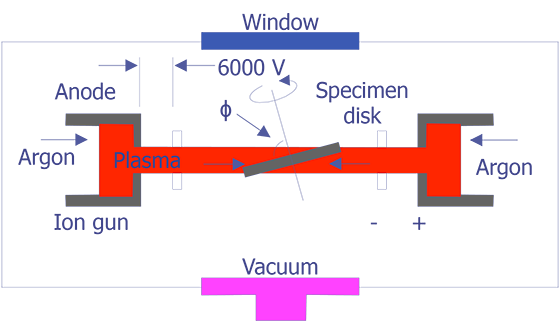
This method is most useful if you need to image a particular point defect, because it yields the best chance for imaging the defect.
Electron energy loss spectroscopy (EELS) is the analysis of the distribution of electron energies emergent from a thin-foil specimen. The electrons in the primary beam are decelerated and lose energy as they pass through the thin sample by several mechanisms, but it is the loss due to ionization of inner shell electrons in the sample that provides the most useful information. The electron spectrometers used for EELS detection are electrostatic or magnetic. The EELS technique is more sensitive to light elements than is EDS; hence, EDS and EELS are often used as complementary techniques.